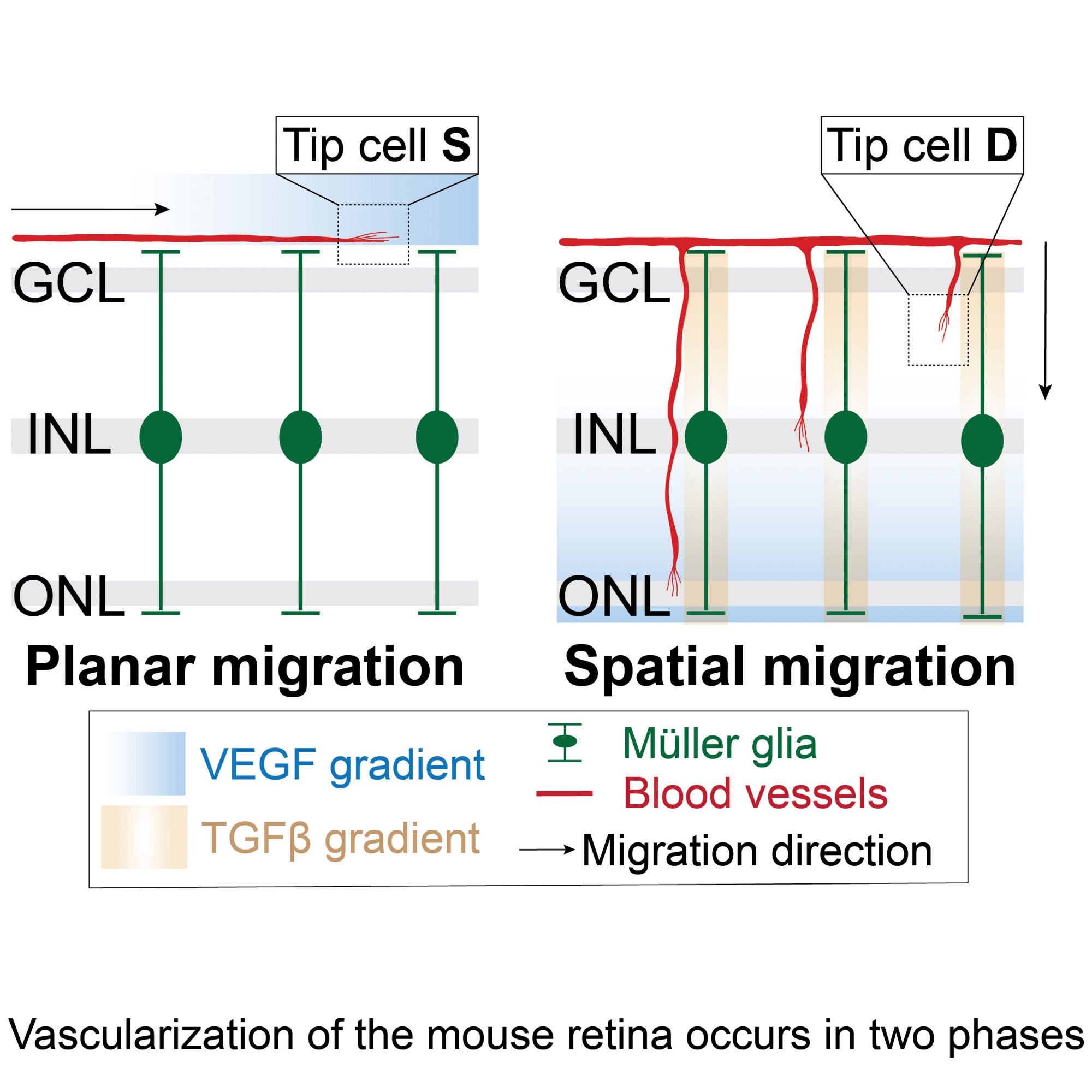
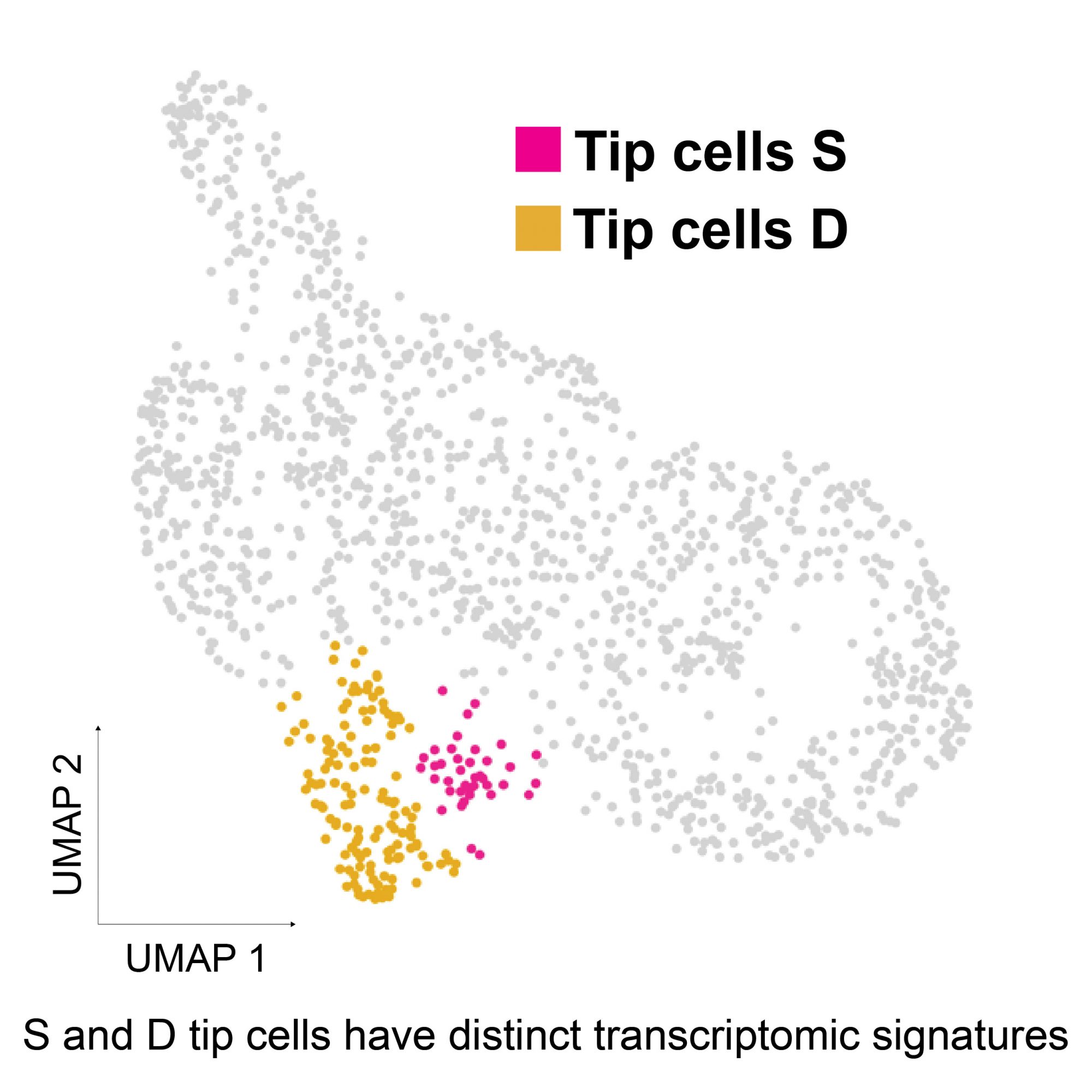
During angiogenic sprouting, endothelial cells adopt two specialized phenotypes; tip cells that spearhead the angiogenic sprout, and stalk cells that follow the tip cells and proliferate to form the vascular lumen. Tip cells have a unique gene expression profile that allows them to migrate and lead the growing sprout. In the lab, we use the mouse postnatal retina as a model to study angiogenic sprouting. During the first postnatal week of retina vascularization, tip cells migrate from the optic stalk on the retinal surface to form the superficial vascular plexus. Once they reach the retinal periphery, vertical sprouts emerge and invade the neuroretina to give rise to the deep vascular layers. Using single-cell RNA sequencing, we compared the identities of superficial vs. diving tip cells and found that diving (D) tip cells have a distinct molecular signature compared to superficial (S) tip cells and acquire blood-retina-barrier (BRB) properties. We are presently studying a number of signaling pathways that are responsible for D-tip cell identity acquisition, and how they affect retinal vascular development. In addition we are investigating how endothelial cell identity changes are involved in the pathogenesis of neovascular diseases, such as retinopathy of prematurity.
Relevant Publications & Media